Porous Ti for Biological Bone Implants
There is a need to develop the next generation of medical implants. Currently, most load bearing bone replacements are bulk metallic implants such as pure titanium and some titanium-based alloys. The application of these materials still suffers from problems such as bone-implant interface instability and biomechanical mismatch of elastic moduli. At Northwestern University, a novel form of porous titanium has been presented as an alternative to alleviate these problems by reducing the stiffness to be comparable to bone stiffness and allowing complete bone ingrowth. However, the porous microstructure causes stress and strain localizations, local permanent plastic strain and residual stress under cyclic loading. My work focuses on the simulation of their mechanical behavior to facilitate the design and development of the porous material. As an alternative to complex tomography procedures, a methodology has been developed to construct a simulated microstructure that retains essential features of the experimental material. The methodology combines stereology theory and the foaming process of the material. A simulated microstructure is shown in Figure 1. 2D and 3D finite element analysis has been performed to predict microscopic stress and strain localizations as well as macroscopic responses. As an example, Figure 2 shows the microstructure of a typical sample and the Von Mises stress field after one loading cycle. The impact of porosity level, pore morphology, and bone-filled pores are being investigated. By the analysis, the porous material will be optimized to most amenable to bone cell ingrowth and have enhanced failure resistance to avoid microstructural failure.
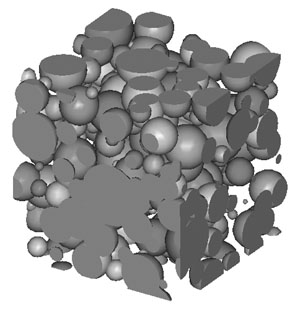

Porous NiTi for Biological Bone Implants
In spite of the advantages mentioned for porous Ti for bone implants (link to other page), issues with strain localization still arise, especially at higher porosity levels desirable for bone ingrowth. A possible solution we are investigating to this problem is design and development of porous nickel-titanium (NiTi) foams. The superelastic and shape memory properties of NiTi alloys include large recoverable strains, thermal actuation possibilities and good damping properties. While NiTi foam inherits the low modulus, light-weight, and improved bone-implant interface of Ti porous materials, the NiTi foam of the same geometry as Ti foam shows no permanent localized strain after cyclic loading as shown in Figure 3. The finite element analysis has been performed by ABAQUS with implementation of a simple perfect pseudoelastic constitutive law. A 3D continuum constitutive model of SMA is under development in Professor Brinson’s group and will be implemented in finite element analysis. The effect of microstructure and bone ingrowth will be studied as well as the stress-strain behavior of the material such as the assessment of damage tolerance under cyclic loading.
Publications:
H. Shen, S. M. Oppenheimer, D. C. Dunand and L. C. Brinson, Numerical Modeling of Pore Size and Distribution in Foamed Titanium, Mechanics of Materials, 2005.
Huanlong Li, Scott M. Oppenheimer, Samuel I. Stupp, David C. Dunand, L. Catherine Brinson, Effects of pore morphology and bone ingrowth on mechanical properties of Microporous Titanium as an Orthopaedic Implant Material, Jap. Materials transactions, Special Issue on "Frontiers of Smart Biomaterials" 45 (4): 1124-1131 (2004).
S. Thelen, F. Barthelat and L. C. Brinson, Mechanics Considerations for Microporous Titanium as an orthopedic implant material, J. Bio Mat. Res., vol. 69A(4), pp. 601-610, (2004).